ADD-ONS / OTHER VASCULAR DISORDERS
Central retinal artery occlusion (CRAO)
Updated on 02/09/2024, published on 01/02/2024
| Ocular vascular disorders |
| Ischemic optic neuropathy (ION) |
|
|
Central retinal vein occlusion (CRVO) → more |
|
|
Hemi-central retinal vein occlusion (HCRVO) |
|
|
Branch retinal vein occlusion (BRVO) |
|
|
Central retinal artery occlusion (CRAO) |
|
|
Branch retinal artery occlusion (BRAO) |
|
|
Cilioretinal artery occlusion |
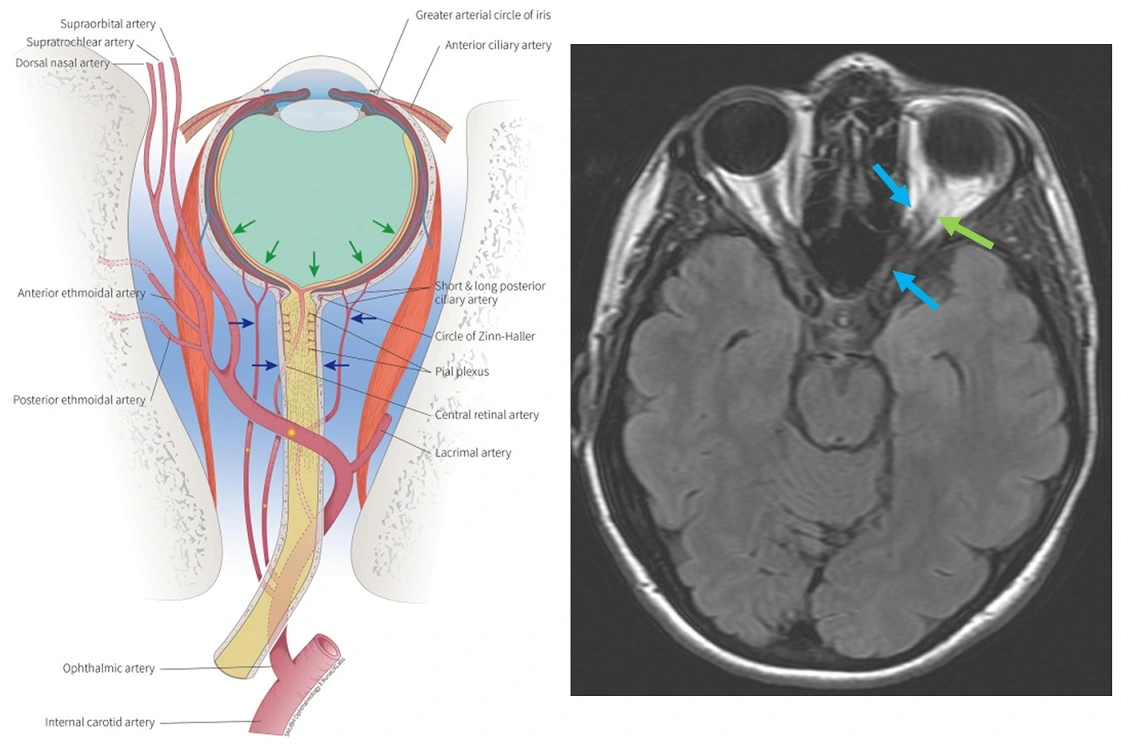
- ophthalmic artery (OA) is the first major branch of the internal carotid artery (ICA), and it usually arises from its supraclinoid segment
- various anomalies of OA origin have been reported (from the cavernous segments of the ICA, MCA, PCoA, or middle meningeal artery)
- OA diameter is ~ 1 mm
- various anomalies of OA origin have been reported (from the cavernous segments of the ICA, MCA, PCoA, or middle meningeal artery)
- OA passes through the dura and enters the orbit via the optic canal, where it runs inferolateral to the optic nerve
- the artery has numerous branches:
- the first branch central retinal artery (CRA) arises just before the ophthalmic artery crosses over the optic nerve; it enters the lower part of the optic nerve and divides into 2 superior and 2 inferior terminal branches
- the OA then crosses over the optic nerve to the medial side and gives off the remaining branches, which may be grouped into those supplying the orbital structures and those supplying the eye; terminal branches are the supratrochlear artery and the dorsal nasal artery
- orbital group
- lacrimal artery (the second and the largest branch)
- supraorbital artery
- posterior ethmoidal artery
- anterior ethmoidal artery
- internal palpebral artery
- supratrochlear artery (frontal artery) and dorsal nasal artery (terminal branches)
- ocular group
- posterior ciliary arteries (including cilioretinal artery) – pass through the sclera and supply the posterior uveal tract
- short posterior ciliary arteries from six to twelve in number
- long posterior ciliary arteries, two for each eye, pierce the posterior part of the sclera a short distance from the optic nerve
- anterior ciliary artery
- muscular branches
- posterior ciliary arteries (including cilioretinal artery) – pass through the sclera and supply the posterior uveal tract
- orbital group
"DR MCLESSI" mnemonic
R = [Central] Retinal Artery
M = Muscular Artery
C = Ciliary Arteries [Long / Short / Anterior]
L = Lacrimal Artery
E = Ethmoidal Arteries [Anterior / Posterior]
S = Supraorbital Artery
S = Supratrochlear Artery [Frontal]
I = Internal Palpebral Artery
- the central retinal artery (CRA) is a branch of the ophthalmic artery (OA)
- the CRA further divides into superior and inferior branches, which eventually branch into nasal and temporal arteries
- in ~ 50% of patients, a cilioretinal artery is present, which can adequately supply the macula even if the CRA becomes occluded
- visual impairment depends on the extent to which the artery supplies the retina; central vision may be preserved in some cases
- central retinal artery occlusion (CRAO) typically leads to acute retinal ischemia with optic nerve edema and necrosis of the optic nerve cells ⇒ severe and permanent visual impairment
- significant spontaneous improvement occurs in <10% of patients
- the incidence of CRAO is reported to be 0.85 per 100,000 people, although the actual numbers are probably higher
Etiopathogenesis
Etiology
- non-arteritic form
- atherosclerosis
- usually > 40 years of age
- ICA stenosis present in up to 45% of CRAOs
- cardioembolism
- angiosclerosis
- vasospasm
- coagulopathy
- polycythemia
- glaucoma
- atherosclerosis
- arteritic form
- most commonly temporal (giant cell) arteritis
- typical in patients > 65 years of age
- most commonly temporal (giant cell) arteritis
|
Occlusion localization
- central retinal artery occlusion (CRAO)
- trunk occlusion leads to complete blindness
- exclude OA occlusion
- branch retinal artery occlusion (BRAO)
- occlusion of either branch results in visual field loss in the corresponding sector
Clinical presentation
- sudden onset, painless, monocular, severe visual disturbance
- patients with a patent cilioretinal artery may have only mild visual impairment
- conservative therapy (without thrombolysis) improves vision in the affected eye in ~ 15% of cases
- occlusion may be transient, presenting as a transient ischemic attack (amaurosis fugax)
Diagnostic evaluation
Ophthalmologic examination
- clinical examination with quantification of visual impairment
- best corrected visual acuity (BCVA)
- reaction to light, motion, counting fingers, etc.
Ophthalmoscopy
- affected part of the retina is pale; the inner retinal layer is absent in the macula, and the choroidal vasculature shines through (cherry-red spot)
- arteries are narrow, filiform, with interrupted blood column
- embolic material may be visible
- ischemic retinal edema resolves within a few weeks, followed by atrophy of the retina and optic disc
Retinal fluorescein angiography (FAG)
- a technique used to examine the circulation of the retina and choroid (parts of the fundus) using a fluorescent dye and a special camera
- detects impaired filling of retinal arteries (delay > 11s or complete absence)
- normal filling of choroidal arteries (complete filling within 5s)
- consider OA occlusion if chorioretinal filling failure is detected
- FAG can assess the effect of recanalization therapy
Electroretinography (ERG)
- ERG is a test that measures the electrical activity of the retina and can be used to diagnose CRAO and assess the severity of the condition
- electrodes are placed on the cornea of the eye, and these electrodes record the electrical signals generated by the retina in response to light stimuli
Etiologic evaluation
- rule out the arteritic form! (→ temporal arteritis)
- assess vascular risk factors and exclude significant carotid stenosis
Blood tests (vascular risk factors)
- CBC, coagulation tests
- erythrocyte sedimentation rate (ESR), CRP
- ionogram, lipid panel, glycemia
- blood cultures (if endocarditis is suspected)
Vascular imaging
- neurosonology / MRA / CTA
- exclude extra-intracranial ICA stenosis
Neuroimaging
- MR-DWI is the best method to exclude clinically silent brain infarcts
- it is better to perform brain CT or MRI (according to local stroke protocol) prior to IVT administration
Other tests
- ECG
- echocardiography (TTE+TEE)
- TCCD bubble test
Management
- spontaneous improvement is rare (< 10-15% of CRAOs)
- none of the conservative treatments (antiplatelet agents, intraocular pressure reduction, vasodilators, eye massage, hemodilution, steroids, heparin) have been proven effective; their use is based on case reports and small series of patients
- intravenous thrombolysis has become the standard of care in eligible patients
- a fundoscopic examination is essential before starting therapy to confirm the diagnosis and to exclude, e.g., intraocular hemorrhage. If arteritis is suspected, ESR and CRP should be obtained (safety of thrombolysis in the arteritic form of CRAO is unknown; in addition, immediate corticotherapy is indicated to prevent occlusion in the fellow eye)
- therapeutic window
- experimental data indicate that retinal cells survive for ~ 120-240 minutes in the presence of complete ischemia
- even with CRA occlusion, complete ischemia rarely occurs; therefore, an interval of 0-12 hours is often used
Conservative treatment
Methods are usually combined, the effect is uncertain, trials have shown no effect compared to placebo [Fraser, 2009]
- mechanical dislodgement of the embolus (repeated bulb massage/ compression)
- vasodilation (IV pentoxifylline, sublingual isosorbide dinitrate)
- intraocular pressure reduction (to increase perfusion pressure in the CRA)
- acetazolamide 500mg IV or PO
- short-term IV mannitol
- methylprednisolone
- anterior chamber paracentesis
- increase blood O2
- oxygen therapy or hyperbaric oxygen therapy (HBOT) within 2-12 hours of onset
Intravenous thrombolysis
- early thrombolysis is more effective than conservative therapy, but randomized trials are lacking [Grory, 2020]
- the therapeutic window is at least 4.5 hours (AHA guidelines 2021) [Grory, 2021]
- one meta-analysis demonstrated safety and efficacy within 4.5 hours [Dumitrascu, 2017]
- “time is retina” – IVT administered within 6.5 has better outcomes than IVT administered in the 6.5-12 hour window [Biousse, 2010] [Hattenbach, 2008]
- the EAGLE trial with a therapeutic window of 20 hours failed to show benefit of IVT
- verify the visual impairment and contraindications to thrombolysis before administering thrombolysis (weigh risk-benefit)
- it is safer to perform brain imaging in all CRAO cases before thrombolysis (to rule out subacute clinically silent ischemia)
-
- (Actilyse) in a standard dosing (AHA Guidelines 2021) [Grory, 2021] [Schultheiss, 2018] [Chen, 2011]
- (Metalyse) may serve as an alternative to alteplase (Vo, 2024)
| Content available only for logged-in subscribers (registration will be available soon) |
Intraarterial thrombolysis
- tPA is usually injected into the ophthalmic artery
- intraarterial tPA dosage is not standardized (alteplase 15-50mg) (AHA guidelines 2021)
- effect of IAT has been reported when started within 15 hours
- the sooner, the better [Aldrich, 2008]
- according to the EAGLE trial, tPA (15-50mg IA) administered within 20 hours had no effect
Secondary prevention
- investigate etiology ASAP ⇒ start individualized stroke prevention (the same as for other types of stroke → Stroke prevention)
- always rule out carotid stenosis, as early CEA may significantly reduce the risk of recurrent stroke