ISCHEMIC STROKE / ACUTE THERAPY
Contraindications to intravenous thrombolysis
David Goldemund M.D.
Updated on 04/06/2024, published on 12/09/2022
Updated on 04/06/2024, published on 12/09/2022
AHA-ASA 2019

Many of the contraindications listed in the 2016 and 2019 guidelines have become relative or obsolete (see comments on IVT)
- mild, non-disabling stroke
-
stroke > 4.5 hours from the onset in unselected patients
-
except for BAO and IVT guided by advanced imaging (see indications for IVT and comments on IVT contraindications)
-
- patients with extensive regions of clear hypoattenuation on CT and extensive DWI lesions (severe stroke) (AHA/ASA 2019 III/A)
- see comments; IVT may be used in selected cases
-
severe head trauma < 3 months
-
use of IV alteplase in patients with a prior ischemic stroke within 3 months (AHA/ASA 2019 III/B-NR)
- relative contraindication; see comments
- recent intracranial or spinal surgery (< 3 months)
- history of ICH
- relative contraindication; see comments
- acute SAH/ICH
- GI tract tumor with high bleeding risk / recent GI bleeding (< 3 weeks) – IVT is potentially harmful
-
coagulopathy
-
platelet count < 100 000/mm3
-
antiplatelet therapy is not a contraindication to IVT (AHA/ASA 2019 I/A I/B-NR)
-
-
known or suspected infective endocarditis (AHA/ASA 2019 III/C-LD)
-
aortic dissection (AHA/ASA 2019 III/C-EO)
-
intra-axial brain tumor – IVT is potentially harmful (AHA/ASA 2019 III/C-EO)
- BP > 185/110 mmHg despite intensive parenteral treatment
- glycemia < 2.7 mmol/l (50 mg/dl) or > 22 mmol/l (400 mg/dl)
- relative contraindication; see comments
Other recommendations
(most relative contraindications are discussed in the comments on IVT)
- Wake-Up Stroke (WUS) – DWI lesion < 1/3 of MCA territory + negative FLAIR (DWI/FLAIR mismatch according to the WAKE-UP trial) (AHA/ASA 2019 IIa/B-R)
-
premorbid disability (including cognitive deficits) – IVT may be considered after evaluation of premorbid status, quality of life, and risk-benefit ratio
- dural puncture in < 7 days – IVT may be considered (AHA/ASA 2019 IIb/C-EO)
- intracranial malformation – the benefits and risks of IVT are unclear; given the increased risk of bleeding, it may be considered in patients with severe deficits if the benefits outweigh the risks
- acute MI – IVT is not contraindicated (AHA/ASA 2019 IIa/C-EO)
- periprocedural stroke – IVT may be considered; prefer direct mechanical thrombectomy ( (dMT) if eligible
-
malignancy – patients with a survival > 6 months and without other malignancy-related contraindications (e.g., coagulopathy, recent surgery, etc.) may benefit from IVT
-
drug-induced stroke – IVT may be administered in the absence of other significant contraindications (AHA/ASA 2019 IIa/C-LD)
-
eye disease – IVT may be used in patients with a history of hemorrhagic retinopathy or other hemorrhagic eye conditions; always weigh the benefits against the risk of complications (AHA/ASA 2019 IIa/B-NR)
- sickle cell disease – IVT may be beneficial (AHA/ASA 2019 IIa/B-NR)
ESO 2021
- new recommendations have been incorporated into the Intravenous thrombolysis (IVT) chapter and the “Comments to some contraindications” tab
- see the full ESO guidelines 2021 here
Comments to some contraindications
- more and more patients, especially in experienced centers, are successfully treated with intravenous thrombolysis (IVT) despite some of the originally published contraindications (off-label thrombolysis)
- this trend is reflected in the recent guidelines and recommendations, where certain contraindications became relative or were abandoned (e.g., advanced age)
- most of the relative contraindications listed in the ESO guidelines 2021 refer to the 0-4.5h time window (with increasing experience, it can be expected to be officially extended to the 4.5-9h window in selected patients)
- typical contraindications that are not respected anymore:
- advanced age
- history of stroke AND diabetes
- 4.5-hour time window
- mild or resolving deficit or, conversely, severe stroke
- aggressive treatment with parenteral antihypertensives to achieve target blood pressure prior to IVT initiation
- use of oral anticoagulants
- always carefully assess the individual risk/benefit ratio
- stroke mimics are associated with a low risk of sICH (→ refer to thrombolysis-related ICH classification for details)
- according to SITS-ISTR, the risk of parenchymal hematoma in stroke mimics was 1.2% [Keselman, 2019]
- according to meta-analyses, the risk of sICH is approx. 0.5% [Tsivgoulis, 2015]
- always exclude isodense SDH ⇒ high risk of bleeding complications!!
- advanced imaging modalities can help confirm true stroke (CTA/MRA, CTP/MRP, DWI)
- seizures are a common stroke mimic but may also occur in the acute phase of a stroke (seizure at the onset of stroke)
- evidence of vascular occlusion on CTA or CTP, or early signs of ischemia on NCCT, may help confirm a stroke diagnosis
- if there is no clear suspicion of mimics or head trauma, IVT is more likely to be performed (ESO guidelines 2021)
- particularly in the case of severe deficit, withholding IVT is likely a greater error than administering thrombolysis to mimics. The risk of thrombolysis-related bleeding in the context of seizure is very low (as it is for all mimics in general) [Polymeris, 2019]
- according to a meta-analysis of the WAKE-UP, EXTEND, THAWS, and ECASS-4 trials, IVT indicated by advanced imaging is safe and effective [Thomalla, 2020]
- if the patient is a candidate for both IVT and MT, IVT is more likely to be performed before MT (consensus) (ESO guidelines 2021)
- IVT can be performed in WUS patients who meet the following criteria (according to guidelines):
- DWI/FLAIR mismatch – DWI lesion < 1/3 of MCA territory + negative FLAIR – meets WAKE-UP trial criteria (AHA/ASA 2019 IIa/B-R) (ESO 2021)
- evidence of penumbra on multimodal CT (EXTEND trial criteria) (ESO guidelines 2021)
- DWI/FLAIR mismatch – DWI lesion < 1/3 of MCA territory + negative FLAIR – meets WAKE-UP trial criteria (AHA/ASA 2019 IIa/B-R) (ESO 2021)
- the ESO 2021 guidelines do not recommend IVT in the 4.5-9 hour window or WUS based solely on NCCT
- this section applies to
- up to 50% of patients with NIHSS 0-6 will have an infarction, and 40% will experience an unfavorable outcome [Strbian, 2013]
- advanced imaging can help decide whether to administer tPA
- in general, a mild, non-disabling deficit should not be thrombolyzed (ESO guidelines 2021) (AHA/ASA 2019 III/B-R)
- see the PRISMS trial results (2018)
- thrombolysis is recommended for patients with:
- mild but disabling deficits (e.g., homonymous hemianopsia, mild aphasia or dysarthria in an actor, mild paresis in a pianist, etc.) (ESO guidelines 2021) (AHA/ASA 2019 I/B-R)
- rapidly improving yet still disabling deficits
- 1/3 of patients will progress and develop a permanent deficit after initial improvement [Smith, 2005] [Smith, 2011]
- potential benefit of IVT (even mild deficits can be bothersome) + low risk of ICH
- mild, non-disabling deficit + persistent cerebral artery occlusion (ESO guidelines 2021)
- neurological deficits may fluctuate, and many patients with acute arterial occlusion will eventually progress if recanalization is not achieved (END – Early Neurological Deterioration)
- however, the TEMPO-2 trial showed no benefit and possible harm from treatment with IV tenecteplase ⇒ patients with minor stroke and intracranial occlusion should not be routinely treated with IVT
- if IVT is not indicated due to symptom resolution, patients should be closely monitored throughout the entire thrombolytic window
- “severe stroke” criteria:
- NIHSS > 25
- presence of early signs of ischemia in > 1/3 of MCA territory
- ASPECTS < 7
- it seems that even in this group of patients, IVT may offer benefits without a concomitant increase in bleeding risk
- a combined analysis of the ECASS, NINDS, and Atlantis trials showed a benefit of IVT but with an increased risk of poor outcome
- the benefit of IVT was evident in patients with NIHSS > 22 [Frank, 2013]
- IVT is recommended for patients with clinically severe stroke lasting < 4.5 hours
- in patients with severe stroke (defined by the extent of early ischemic changes on CT) lasting <4.5 h, IVT should be considered in selected cases (ESO 2021)
- if bleeding risk is further increased (extensive leukoencephalopathy, known microbleeds, etc.), consider direct MT
- previous ICH
- patients with a history of ICH were excluded from thrombolytic trials, and IVT is generally not recommended for this group. Existing data are derived from case series and small non-randomized cohorts, showing no marked increase in ICH incidence [Zhao, 2019]
- ICH risk varies depending on the etiology of the previous bleeding
- amyloid angiopathy or untreated vascular malformations carry a high risk
- hypertensive bleeding > 12 months ago has a relatively low risk
- amyloid angiopathy or untreated vascular malformations carry a high risk
- the decision on IVT is individual (ESO guidelines 2021 – expert consensus) (ESO guidelines 2021 – expert consensus)
- IVT may be recommended if there has been a long period since the bleeding, non-recent subcortical ICH, bleeding from a secure source, or bleeding due to anticoagulant overdose, etc.
- cerebral microbleeds (CMBs)
- an increased risk of bleeding with CMBs > 10 (OR 13 !) ⇒ IVT is not recommended ( (ESO guidelines 2021) [Zand, 2017] [Fiehler, 2007]
- for those with unknown or low cerebral microbleed burden (e.g., <10), IVT is suggested (ESO guidelines 2021)
- probability of finding > 10 CMBs is approx. 0.6-2.7% – MRI screening to assess cerebral microbleeds before making a treatment decision regarding IVT is not recommended
- probability of finding > 10 CMBs is approx. 0.6-2.7% – MRI screening to assess cerebral microbleeds before making a treatment decision regarding IVT is not recommended
- TIA
- data from case reports and small patient series
- patients with a recent TIA (<1 month) do not appear to be at a higher risk of bleeding complications or adverse outcome after IVT [De Leciñena, 2012]
- IVT appears to be safe even in patients with very recent TIA episodes [Tsivgoulis, 2014]
- recent stroke
- IVT can be used in selected cases (e.g., lacunar infarction, infarction occurring > 1 month ago with subsequent good recovery) (ESO guidelines 2021)
- a meta-analysis of 900 patients found no increased incidence of sICH or worse outcome [Tsivgoulis, 2019]
- higher ICH risk is seen in recent strokes (within 14 days); the location and extent of ischemia and the etiology of the previous stroke should also be considered (higher risk of sICH in cardioembolic stroke) [Tan, 2014]
- IVT appears to be safe for asymptomatic infarcts detected on DWI [Stösser, 2020]
- IVT is recommended for patients with an initial BP >185/110 mmHg that has been lowered to <185 and <110 mmHg (with/without antihypertensive drugs) (ESO guidelines 2021)
- a subanalysis of the IST-3 trial and other smaller studies have not shown an increased risk of bleeding in patients treated aggressively for hypertension [Martin-Schild, 2008]
- stringent BP control is required for 24h after thrombolysis
- another analysis showed a higher incidence of sICH in patients with BP > 185/110; however, they had a better clinical outcome (mRS) compared to controls [Frank, 2013]
- intravenous thrombolysis is not recommended for patients with persistently elevated BP >185/110 mmHg despite antihypertensive treatment (ESO guidelines 2021)
- originally, IVT was contraindicated for hypoglycemia (< 2.7 mmol/l / 50 mg/dL) or hyperglycemia ( > 22 mmol/L / 400 mg/dL)
- hypoglycemia, in particular, can lead to “stroke mimics”
- hyperglycemia is associated with a worse outcome and ↑ risk of sICH
- according to expert recommendations:
- no contraindication in case of confirmed stroke (CT/CTA/CTP or MR-DWI)
- IVT is suggested in the presence of glycemia > 22 mmol/l (400 mg/dl) even without evidence of arterial occlusion (ESO guidelines 2021)
- if hypoglycemia is corrected and deficit persists, IVT may be performed (administration of IVT in patients with stroke mimics is relatively safe)
- IVT should not interfere with the administration of insulin therapy in acute stroke patients with high blood glucose
- IVT is not recommended if the platelet count is < 100 000 /mm3 (ESO guidelines 2021)
- IVT may be considered (off-label) if the platelet count is between 70 000-100 000 /mm3 [Tsivgoulis, 2021]
- if the platelet count is unknown and there is no reason to expect abnormal values, IVT may be started while awaiting laboratory results (ESO guidelines 2021)
- DAPT is associated with only a modest increase in ICH and does not worsen outcome [Malhotra, 2020]
- mortality 17.9% vs. 16.6%, sICH 2.9% vs. 1.5% (according to SITS-MOST criteria) [Tsivgoulis, 2018]
- IVT is not contraindicated in patients on single or dual antiplatelet therapy (AHA/ASA 2019 I/B-NR) (ESO guidelines 2021)
- intra-axial tumors remain a contraindication to IVT due to the high risk of sICH
- patients with extra-axial tumors (typically meningiomas) can be treated with IVT after an individualized risk-benefit assessment
- optimally, an MRI should be performed to detect microbleeds in the tumor and to assess the extent of collateral edema
- renal insufficiency is generally not a contraindication to IVT
- severe CKD (<30 mL/min per 1.73 m2) is associated with higher mortality and worse outcome, but not with increased sICH; it may be a consequence of comorbidities rather than IVT itself
- avoid IVT in cases of unfavorable risk-benefit ratio (e.g., minor stroke + poorly controlled hypertension or multiple relative contraindications)
- APTT must be within the normal range
- IVT appears to be safe for saccular aneurysms, including larger ones (> 10 mm) [Virta, 2021]
- IVT can be performed in patients with unruptured aneurysms (without specifying the size) (ESO guidelines 2021)
- patients with AVMs, cavernomas, and DAVFs should be treated individually – data are limited
- according to older European recommendations, the interval between IVT and surgery should be 3 months; according to AHA/ASA 14 days, new ESO guidelines 2021 recommend 14 days
- IVT may be safely performed in postoperative patients as an off-label procedure; risk-benefit should be evaluated based on the type of surgery and the compressibility of the surgical field
- in a cohort of patients thrombolyzed after previous surgery, including major procedures, the risk of bleeding was 7%, of which 3% were major; the risk was higher for recent surgery (< 10 days, OR 10) [Voelkel, 2017]
- a relative contraindication
- according to available data, it is better not to administer IVT in case of recent GIT bleeding (< 3 weeks) and severe traumatic brain injury (TBI) in < 3 months (consider the type and extent of injury and CT findings)
- recent extracranial trauma (< 14 days) – IVT may be considered (AHA/ASA 2019)
- isolated extracranial carotid dissection – IVT recommended (ESO guidelines 2021)
- aortic dissection – absolute contraindication to IVT! (ESO guidelines 2021)
- intracranial dissection – IVT should be avoided (expert consensus) (ESO guidelines 2021)
- even a high burden of white matter lesions is not a clear contraindication to IVT within 4.5 hours (ESO guidelines 2021)
- acute myocardial infarction (MI) (< 6h) is not a contraindication to IVT (standard tPA dose 0.9 mg/kg is used)
- subacute MI (> 6h – 7 days) with ST elevation – IVT is not recommended (ESO guidelines 2021)
- subacute MI (>1 week and < 3 months) was the official contraindication to IVT (↑ risk of hemopericardium and tamponade); however, according to AHA/ASA guidelines 2018 and ESO guidelines 2021, IVT may be performed in non-STEMI and considered in STEMI of inferior wall, right heart and left anterior wall (ESO guidelines 2021)
- most complications occur in recent STEMI ( < 1 week from onset)
- the extent of myocardial infarction and available TTE findings should be considered
- most complications occur in recent STEMI ( < 1 week from onset)
- infective endocarditis – IVT is not recommended in patients with a definite or suspected diagnosis of infective endocarditis (ESO guidelines 2021)
- pericarditis – IVT may be considered in severe stroke; the benefit for mild stroke is unclear; consult a cardiologist
-
intracardiac thrombus – IVT should be considered in severe stroke; the benefit in mild stroke is unclear
-
myxoma and fibroelastoma – IVT should be considered for severe stroke; the benefit in mild stroke is unclear
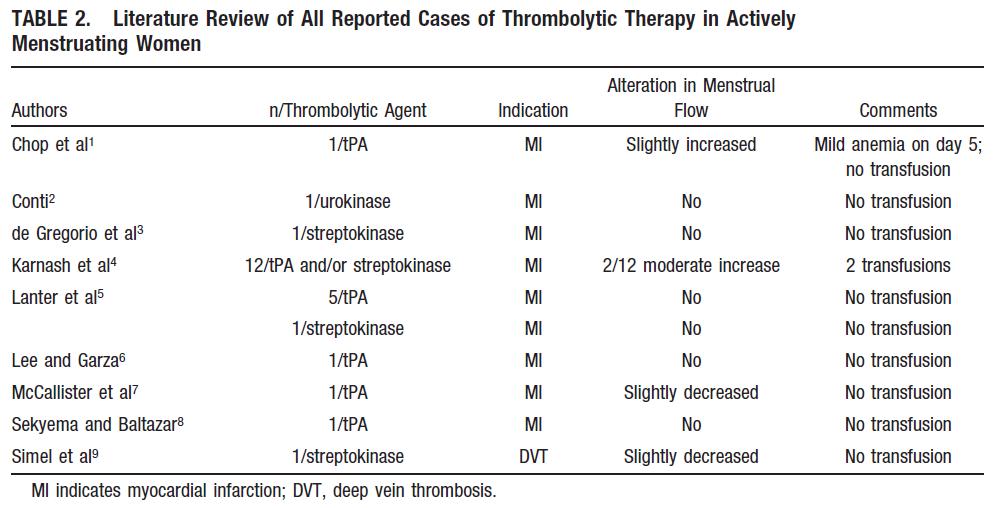
Menstruation
- according to expert consensus, women experiencing an acute stroke during menstruation who otherwise meet eligibility criteria may be treated with IVT on an individual basis after appropriate evaluation of the risk-benefit profile (ESO 2022) (AHA/ASA 2019 IIa/C-EO)
- the risk of bleeding peaks in the first 20 hours, when platelets and fibrin are the primary hemostatic factors
- after 20 hours, arteriolar constriction in the basal endometrial layer and myometrial contraction predominate in hemostasis, and there is no dependence on clot formation
- benefits of IVT probably outweigh the risk of significant bleeding
- the risk of bleeding peaks in the first 20 hours, when platelets and fibrin are the primary hemostatic factors
- IVT may be considered in patients with a history of recent menorrhagia without clinically significant anemia/hypotension (AHA/ASA 2019 IIa/C-EO)
- consultation with a gynecologist is advised for patients with recent or ongoing vaginal bleeding accompanied by anemia (a (AHA/ASA 2019 IIa/C-EO)
- patients should be informed of the potential need for transfusion.
- direct MT may be considered in patients with large vessel occlusion (LVO)
- extensive data from clinical trials are lacking; only case reports (usually with uncomplicated courses) have been published [Sekyema, 1995]
- the NINDS trial enrolled 9 menstruating women, 5 of whom were actively treated. Severe bleeding requiring transfusion and subsequent elective uterine artery embolization occurred in 1 patient with a 1-year history of dysfunctional vaginal bleeding [Wein, 2002]
- in the GUSTO-I trial, 12 patients received either streptokinase or tPA for acute MI. None experienced severe bleeding; 3 patients had moderate vaginal bleeding (2 requiring transfusion) [Carnash, 1995]
Gravidity
Intravenous thrombolysis (IVT)
- due to its large molecule size, tPA does not cross the placental barrier; there is no evidence of teratogenicity
- there are concerns about placental abruption, intrauterine hemorrhage, preterm delivery, or fetal death
- there are no data from RCTs (pregnancy was an exclusion criterion)
- most case reports and small series are from patients who received thrombolysis for non-stroke diagnoses rather than stroke (PE, valvular thrombosis, DVT, stroke, myocardial infarction)
- in 8 published cases of IVT, only 1 case of uterine bleeding occurred
- according to a published cohort of 28 patients, the risk of TL-related abortion was 8%, and the maternal risk of IVT did not differ from that of non-pregnant patients [Leonhardt, 2006]
- case reports of successful IVT for stroke in late pregnancy have also been published [Wiese, 2006] [Daprich, 2006]
- all recommendations are based on expert consensus (ESO guidelines 2022)
- IVT may be considered on an individual basis for moderate and especially severe stroke when the expected benefit of IVT outweighs the risk of uterine bleeding (ESO guidelines 2022 – expert consensus) (AHA/ASA 2019 IIb/C-LD)
- factors favoring IVT:
- severe, debilitating deficit
- likely benefit of IVT
- MR DWI/PWI mismatch and small lesion on DWI
- likely peripheral occlusion – from M2 distally
- if MT is available and LVO is involved, direct MT is preferable to bridging therapy
- stroke occurring shortly after delivery
- the safety and efficacy of IVT shortly after delivery (<10 days) are not established (AHA/ASA 2019 IIb/C-LD)
- if the stroke occurs > 10 days after delivery, IVT may be given after individual consideration (ESO guidelines 2022 – expert consensus)
if MT is available and there is a presumption of increased bleeding risk, dMT is preferred (ESO guidelines 2022 – expert consensus)
Mechanical recanalization
- there is relatively limited experience with MT in pregnant women; no RCTs are available
- direct MT may be preferable to bridging therapy for LVO during pregnancy and shortly after delivery
- fetal shielding with lead vests is required
IVT in children
| Content available only for logged-in subscribers (registration will be available soon) |