OTHER VASCULAR DISORDERS
Aortic dissection
David Goldemund M.D.
Updated on 07/11/2023, published on 05/05/2023
- the aorta is the main artery of the human body, divided by the diaphragm into the thoracic and abdominal segments
- in healthy adults, the aorta is usually less than 40 mm in diameter and gradually narrows downwards
- aorta increases its diameter by approx. 0.9 mm in men and 0.7 mm in women every 10 years
- aorta increases its diameter by approx. 0.9 mm in men and 0.7 mm in women every 10 years
- aortic dissection is characterized by a tear in the intimal layer of the aorta, allowing blood to enter and dissect the layers of the wall
- a relatively rare disorder with a high mortality rate; it is the most common form of acute aortic syndrome (AAS)
- AAS = a group of aortic emergencies with similar clinical characteristics, including intramural hematoma, penetrating aortic ulceration, dissection, and rupture
- aortic dissection remains one of the few absolute contraindications to intravenous thrombolysis
 Definition and classification
Definition and classification
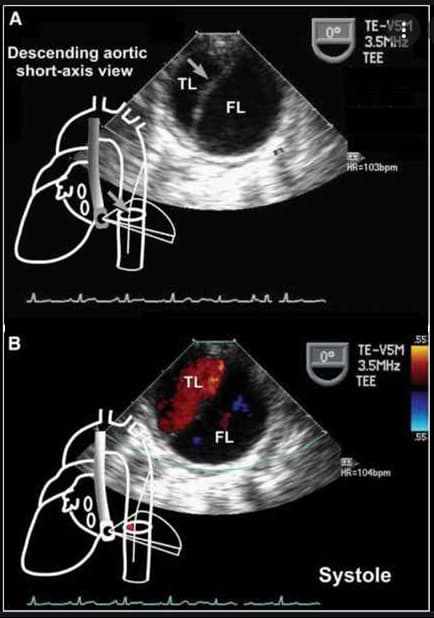
- an intimal tear (or ulcer penetration) leads to bleeding into the medial layer of the aortic wall, subsequently forming a true and false lumen
- the true lumen is lined by the intima; the false lumen is a blood-filled channel within the medial layer
- the process may be followed by aortic rupture or reentry into the true lumen via a secondary intimal tear (reentry point)
- main complications:
- cardiac tamponade
- aortic regurgitation
- proximal or distal malperfusion syndromes (periphery, CNS)
- classifications
- DeBakey
- type I – begins in the ascending aorta and extends to the abdominal segment
- type II – dissection is limited to the ascending aorta
- type III – dissection originates in the aortic isthmus and involves the descending aorta, potentially extending to the abdominal segment
- Stanford classification
- type A – the ascending aorta is affected (twice as common as type B) (∼ 60%)
- type B – the descending aorta is affected
- classification based on timing
- acute: ≤ 14 days from symptom onset
- subacute: 14 days – 3 months
- chronic: > 3 months from symptom onset
- communicating x non-communicating
- DISSECT classification (Duration, Intimal tear, Size of the dissected aorta, Segmental extent of involvement, Clinical complications, Thrombosis of the false lumen)
- DeBakey
 Etiology
Etiology
- a complication of an underlying inherited connective tissue disorder
- Marfan syndrome
- Ehlers-Danlos syndrome
- Loeys-Dietz syndrome
- idiopathic (with some known predisposing factors)
- atherosclerosis (penetrating ulcer)
- hypertension (medial degeneration)
- vasculitis
- structural aortic abnormalities (bicuspid aortic valve, aortic coarctation)
- pregnancy
- previous intervention (cardiac or aortic surgery, intra-aortic balloon pump)
- use of fluoroquinolones
 Epidemiology
Epidemiology
- incidence 6/100,000/year
- typically occurs in elderly hypertensive patients; higher incidence in men than in women
- up to 38% of cases remain undiagnosed at the initial examination
- prognosis is worse in women due to atypical symptoms and delayed diagnosis
 Clinical presentation
Clinical presentation
- chest pain (70-80%) or back pain (more common in type B)
- the most typical symptom; however, about 6% of patients are completely pain-free
- sudden onset of severe, shooting pain in the chest and/or back
- sometimes intermittent or migrating
- aortic regurgitation (40-70%) – dissection leads to dilatation of the aortic root and aortic annulus, tearing of the aortic cusps; a diastolic murmur over the aorta is clinically present
- hypotension or signs of shock (25% of patients)
- may result from aortic rupture, acute significant aortic regurgitation, massive myocardial infarction, cardiac tamponade, or major blood loss
- myocardial ischemia (10-15%)
- congestive heart failure
- syncope (type A 15%, type B < 5%)
- evidence of malperfusion (depending on the extent of dissection and occlusion of aortic branches)
- neurologic symptoms
- stroke (< 5-10%)
- spinal ischemia (< 1%) due to involvement of the artery of Adamkiewicz
- abdominal ischemia (mesenteric artery)
- limb ischemia (different BP in upper and lower extremities, weak pulsations)
- renal failure – 20% of patients with acute type A and approx. 10% of patients with type B
- neurologic symptoms
 Diagnostic evaluation
Diagnostic evaluation
- diagnostic procedures aim to confirm or exclude dissection; their choice depends on the risk assessed by the medical history and clinical presentation (ADD-RS)
- D-dimer testing is advised only in stable, low-risk patients (not recommended in cases with a high clinical probability of aortic dissection)
- with a low clinical probability of AAS, negative D-dimers should be considered as an exclusion of AAS
- intermediate clinical probability of AAS together with a positive D-dimer result warrants further imaging studies
- transthoracic echocardiography (TTE) is recommended as the initial imaging, followed by:
- computed tomography (CT) or transesophageal echocardiography (TEE) in unstable patients
- CT, TEE, or MRI in stable patients
- if initial imaging is negative, reimaging (CT/MR) is recommended if AAS is still suspected
- for uncomplicated type B dissection managed conservatively, reimaging (CT/MR) within 24 hours is recommended
| Clinical data relevant for assessing the initial probability of acute aortic syndrome (AAS) Aortic Dissection Detection Risk Score (ADD-RS) |
||
| high-risk medical history data (1 point for any) |
high-risk pain characteristics (1 point for any) |
high-risk clinical findings (1 point for any) |
|
|
|
- ADD-RS is a clinical tool for stratifying the likelihood of aortic dissection (low risk = 0 points, moderate risk = 1, high risk = 2-3)
- a score of 0 points on ADD-RS + negative D-dimer provide a relatively reliable method for ruling out acute aortic syndrome
Clinical examination and laboratory test
- elevated D-dimer levels increase the probability of aortic dissection (at the same time, rule out pulmonary embolism!)
- rapid increase ⇒ high diagnostic value in the first hours
- negative DD in low-risk patients = aortic dissection is excluded
| CBC | anemia |
| leukocytosis | inflammation |
| C-reactive protein | inflammation |
| procalcitonin | DDx of SIRS and sepsis |
| creatine kinase (CK) |
reperfusion injury, rhabdomyolysis |
| troponin I or T | ischemia/myocardial infarction |
| D-dimers | aortic dissection, pulmonary embolism, thrombosis |
| creatinin | renal failure (existing or developing) |
| AST, ALT | liver ischemia, liver disease |
| lactate | intestinal ischemia, metabolic disorders |
| glucose | diabetes mellitus |
| ASTRUP |
metabolic and oxygenation disorders |
Imaging methods
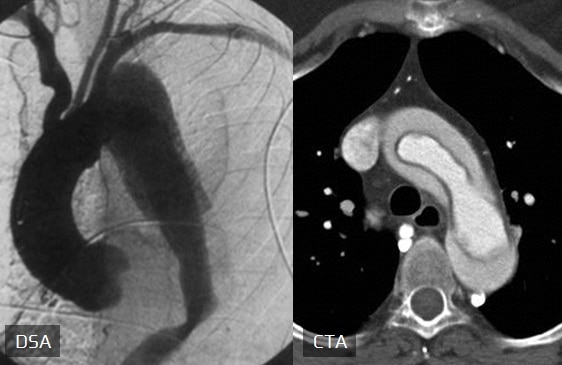
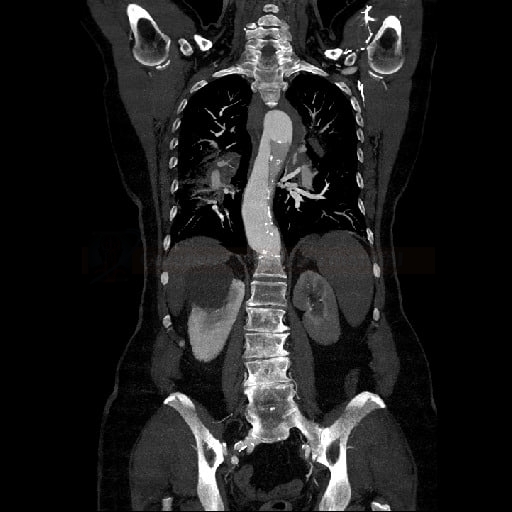
CT scan, MRI and TEE have comparable reliability in confirming or excluding the diagnosis of acute aortic dissection
Findings to be reported in case of aortic dissection
- evidence of intimal flap
- assessment of the proximal and distal extent of dissection (which aortic segments are involved) + aortic size
- identification of false and true lumen (+ presence of thrombosis)
- location of the intimal tear (entry and potential reentry)
- identification and degree of aortic regurgitation
- involvement of aortic branches
- detection of impaired perfusion (characterized by reduced or absent flow)
- detection of organ ischemia or vascular occlusion (e.g., brain, myocardium, bowel, kidneys, etc.)
- detection and extent of pleural or pericardial effusion
- signs of mediastinal and periaortic bleeding
 Differencial diagnosis
Differencial diagnosis
- pseudodissection caused by the aortic pulsations (use ECG-gated CTA to avoid this motion artifact)
- “pseudo flap” appearance on some MRI sequences (due to susceptibility artifact)
- mural thrombus
- intramural hematoma, penetrating atherosclerotic ulcer (both part of an acute aortic syndrome)
 Management
Management
- pharmacotherapy for pain control and blood pressure optimization is recommended for all patients with aortic dissection (ESC 2014 I/C)
- target SBP < 100-120 mm Hg
- target HR < 60/min
- beta-blockers are preferred
- urgent surgical treatment is recommended for patients with aortic dissection type A (ESC 2014 I/B)
- in patients with type A + malperfusion, a hybrid approach should be considered (i.e., replacement of the ascending aorta and/or aortic arch combined with any percutaneous intervention on the aorta or outflowing arteries) (ESC 2014 IIa/B)
- in patients with uncomplicated type B dissection, pharmacotherapy is recommended (ESC 2014 I/C)
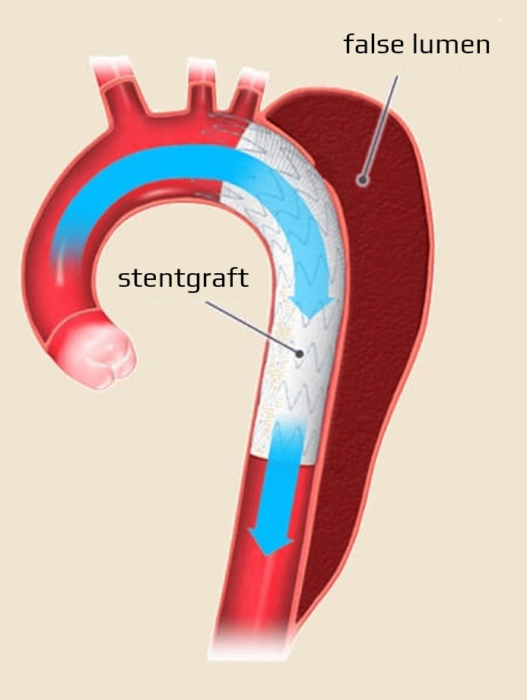
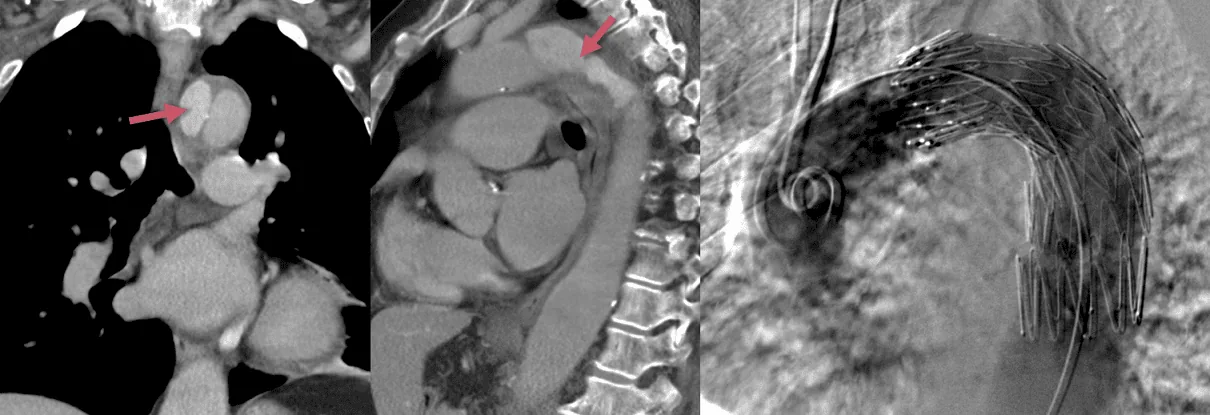
- consider TEVAR (thoracic endovascular aortic repair) in patients with uncomplicated type B (ESC 2014 IIa/B)
- TEVAR is recommended in patients with complicated type B (ESC 2014 I/C)
- surgical treatment may be considered in patients with complicated type B (ESC 2014 IIb/C)
Aortic dissection type A
- surgery is the treatment of choice
- patients with acute type A have a 50% mortality rate within the first 48 hours without surgical intervention
- high perioperative mortality (25%) and incidence of neurological complications (18%) persist despite advancements in surgical techniques and anesthetic management
- although neurological deficits or coma upon admission are associated with poor postoperative prognosis, restitution of neurological findings has been described in some cases where cerebral perfusion was rapidly restored (especially if the interval from symptom onset to surgical intervention was < 5 hours)
Aortic dissection type B
- the clinical course of type B dissection is often uncomplicated
- in the absence of signs of malperfusion or disease progression, the patient may be safely stabilized with medical therapy alone
 Complications
Complications
Type A and B
- malperfusion due to:
- dissection with stenosis/occlusion of aortic branches
- distal thromboembolism
- aneurysmal dilatation
- aortic rupture
- hemothorax
Stanford type A specific complications
- coronary artery occlusion with MI
- aortic incompetence
- aortic valve insufficiency
- hemopericardium with cardiac tamponade
- hemorrhagic shock