ISCHEMIC STROKE / COMPLICATIONS
Acute ischemic stroke complications
Updated on 21/05/2024, published on 20/02/2023
- typically, the neurological deficit is maximal at the onset of the ischemic stroke and may subsequently improve (either spontaneously or due to recanalization therapy)
- however, stroke patients are at risk of a variety of complications that may lead to death or clinical deterioration, manifested by:
- progression of existing deficits or appearance of new symptoms (due to new occlusion, progression of existing thrombosis, edema, or hemorrhage)
- quantitative or qualitative impairment of consciousness
- delirium
- epileptic seizures
- intracranial hypertension
- mood disorders
- worsening of preexisting comorbidities (cardiac, respiratory, etc.)
- onset of new systemic disorders (infection, venous thromboembolism, infarction, etc.)
- complications can be divided into intracranial and extracranial; a brief overview follows
Diagnostic evaluation
- general and neurological examination
- review medication (sedatives, delirium-inducing drugs)
- follow-up CT/MR of the brain (large edema? hemorrhage? new infarct lesion? hydrocephalus?)
- follow-up vascular imaging (neurosonography/MRA/CTA)
- new occlusion in another segment? reocclusion of recanalized artery?
- hemodynamically significant stenosis?
- collateral circulation failure?
- EEG examination (or EEG monitoring)
- ECG monitoring
- other imaging studies
- chest X-ray
- abdominal ultrasound
- CT scan of the chest, abdomen, or pelvis
- laboratory tests:
- basic metabolic panel, CBC + coagulation tests
- arterial blood gas (ABG) analysis (arterial pH, PaO2, PaCO2, and “base excess”
- test provides valuable information about the patient’s acid-base balance, oxygenation, and ventilation status, which are crucial in the management of stroke and other critical conditions
- normal arterial pH: 7.35-7.45
- CRP, procalcitonin
- cardiac enzymes to assess the myocardial injury
- troponin T (TnT) and troponin I (TnI)
- creatine kinase-MB (CK-MB)
- Mg, Ca, ammonia
- basic metabolic panel, CBC + coagulation tests
- detection of infection
- sputum and urine analysis, culture swabs
- blood cultures
Intracerebral complications
- approximately 30% of acute stroke patients experience progression of neurological deficits, including quantitative or qualitative disturbances of consciousness
- deterioration may be gradual (typically due to edema or thrombus progression) or sudden (new embolism, abrupt failure of collateral circulation, or intracranial bleeding, etc)
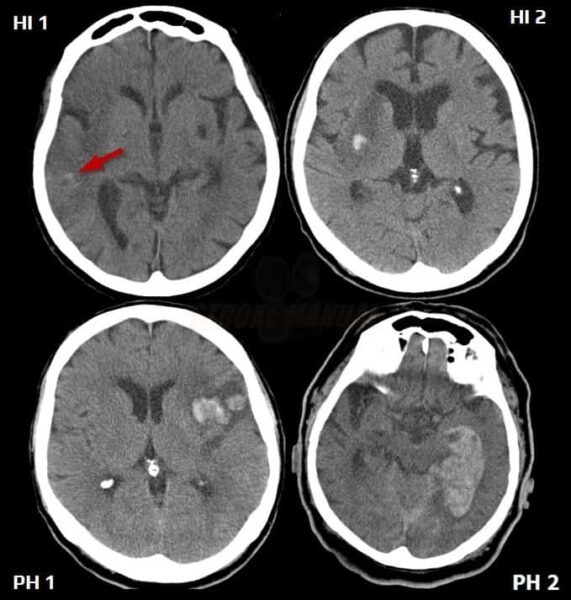
Hemorrhagic transformation of ischemia
- the hemorrhagic component is easily detected on CT and even better on MR GRE
[Arnauld, 2003]
- hemorrhagic transformation most commonly occurs within the first 48 hours; increased risk is associated with extensive ischemia, late recanalization, and early anticoagulation
- hemorrhagic transformation is often asymptomatic
- antiplatelet therapy/LMWH should be discontinued if major bleeding occurs
Brain edema, intracranial hypertension
- edema usually develops between days 2-5
- initially intracellular (cytotoxic)
- a vasogenic component can be seen from day 5
- symptomatic edema can be expected in large supratentorial ischemias and cerebellar infarcts
- clinical presentation:
- worsening of the neurological deficit (including loss of consciousness) within 24-36 hours
- gradual progression over several days
- initial deterioration is followed by a plateau and gradual improvement over a week (except for malignant edema cases)
- worsening of the neurological deficit (including loss of consciousness) within 24-36 hours
- efficacy of pharmacotherapy is unclear (AHA/ASA 2013 IIb/C)
- ventricular drainage and/or craniectomy is recommended if acute hydrocephalus develops as a result of a posterior fossa stroke (AHA/ASA 2013 I/C)
- decisions regarding decompressive craniectomy should be made early in cases of malignant ischemia
Ischemia progression / early stroke recurrence
| Content available only for logged-in subscribers (registration will be available soon) |
Acute symptomatic epileptic seizures (ASS)
- according to various sources, stroke is responsible for up to 60% of all acute symptomatic seizures (ASS)
- seizures are typically focal, with possible secondary generalization
- most seizures occur in the first 48 hours after stroke onset; increased risk of ASS is associated with large cortical ischemia and intracerebral bleeding
Delirium
- delirium (historically known as acute confusional state) presents with disturbances in attention, awareness, and higher-order cognition
- commonly attributable to:
- brain lesion
- extracerebral causes: medication, metabolic disorders, alcohol withdrawal syndrome (AWS), infection, etc.
- Alcohol Withdrawal Syndrome (AWS) is a set of symptoms that can occur following a reduction in alcohol consumption after a period of excessive and/or prolonged use (besides delirium, tremor, anxiety, nausea, hallucinations, or seizures can be seen)
- Alcohol Withdrawal Syndrome (AWS) is a set of symptoms that can occur following a reduction in alcohol consumption after a period of excessive and/or prolonged use (besides delirium, tremor, anxiety, nausea, hallucinations, or seizures can be seen)
- combination of both intracerebral and extracerebral factors
| Delirium-inducing drugs |
|
anticholinergic drugs
|
|
|
Addictive substances and alcohol (acute toxicity/ withdrawal)
|
|
H2-blockers (cimetidine, ranitidine)
|
|
opioid analgesics
|
|
antimalarial drugs (mefloquine)
|
|
antiviral drugs (acyclovir)
|
|
others (lithium, barbiturates, benzodiazepines flunitrazepam, NSAIDs indomethacin, digoxin, corticosteroids, centrally acting myorelaxants, antihypertensives
|
Failure of collateral circulation
- the risk of collateral circulation failure is increased in the presence of concurrent extra-intracranial stenoses and/or hypotension
- typical watershed infarcts can be seen in such settings
- maintain a mean arterial pressure (MAP) >110 mmHg in normotensive patients and >130 mmHg in patients with known hypertension for the first 24 hours
- administer plasma expanders or vasopressors if needed
Extracerebral complications
Infectious complications
- fever is most commonly caused by respiratory infections
- preventive measures for aspiration bronchopneumonia include early detection of dysphagia and early indication for NG tube placement
- preventive measures for aspiration bronchopneumonia include early detection of dysphagia and early indication for NG tube placement
- urinary tract infections are associated with catheter use
- catheter-related bloodstream infections (→ sepsis)
Metabolic disorders
- hydration disorders (usually dehydration)
- ionic imbalances (most commonly hyponatremia, hypokalemia, hypophosphatemia, etc.)
- renal or prerenal uremia
Cardiovascular complications
- several heart conditions (such as valvular heart disease, atrial fibrillation, and myocardial infarction) are significant risk factors for stroke
- acute stroke can also induce cardiac dysfunction (e.g., arrhythmias) or lead to other cardiac complications
- worsening of preexisting cardiomyopathy (CMP) or congestive heart failure (CHF)
- a risk factor for increased morbidity and mortality
- acute decompensation may occur due to myocardial infarction (MI), tachyarrhythmias, transfusion, or antiedema therapy
- acute stress cardiomyopathy (Tako-Tsubo)
- characterized by bulging of the apical part of the ventricle, ST-segment elevation with negative T waves in leads V3 and V4
- cardiac enzymes are typically normal
- females are more commonly affected
- myocardial infarction with hypokinesia may be a source of cardiac embolism, but MI may also be a consequence of stroke (autonomic dysregulation, stress)
- elevation of cardiac enzymes due to neurogenic myocardial damage (myocytolysis) is a frequent finding; exclusion of MI is imperative
- more common in polymorbid patients with previously established coronary artery disease (CAD) and vascular risk factors (such as diabetes, hyperlipidemia, etc.)
- decompensated hypertension or hypertensive crisis (emergency) occurs in up to 80% of stroke patients
- etiology is complex, involving compensatory mechanisms aimed at improving perfusion and stress response
- hypotension is generally defined as SBP < 90 mm Hg or DBP < 60 mm Hg
- the clinical threshold (leading to hypoperfusion symptoms including border zone infarction) is individual (higher in hypertensive patients)
- most common causes of hypotension in acute stroke patients are:
- heart failure
- hypovolemia/shock
- anemia
- sepsis
- arrhythmias (dysrhythmias) are irregularities in heart rhythm and are a common complication of acute stroke
- the most common are supraventricular arrhythmias
- sinus tachycardia
- atrial fibrillation/flutter
- arrhythmias may cause:
- hemodynamic instability with hypotension (↑risk of hypoperfusion injury)
- congestive heart failure, potentially leading to pulmonary edema, etc.
- atrial fibrillation/flutter increases the risk of cardioembolism
Respiratory complications
- hypoxia leads to a progression of brain damage, transitioning from penumbra to necrosis and potential worsening of neurological deficit
- the most common causes of respiratory failure are:
- impaired airway clearance
- congestion, difficulty coughing (due to bulbar syndrome or somnolence) ⇒ hypoxemia, hypercapnia
- airway obstruction
- aspiration pneumonia
- often a combination of aspiration and congestion/difficulty in coughing
- increased risk in drowsy patients and those with bulbar syndrome
- early screening for dysphagia may prevent pneumonia
- central respiratory disorders (due to primary and secondary brainstem lesions)
- pulmonary edema, atelectasis
- pulmonary embolism
- decompensation of asthma bronchiale (caution when using beta-blockers)
- impaired airway clearance
Gastrointestinal complications
- dysphagia is present in 42-67% of patients with acute stroke in the first three days
- the most severe dysphagia is seen in patients with brainstem lesions
- dysphagia is associated with an increased risk of aspiration
- the incidence of aspiration in the first five days ranges from 20% to 42%
- aspiration/silent aspiration is a frequent cause of bronchopneumonia
- systematic screening for dysphagia reduces the risk of aspiration-related bronchopneumonia
- recommendations:
- screening for dysphagia before initiating per os intake (including medications) – effective in identifying patients at increased risk of aspiration (AHA/ASA 2019 I/C-LD)
- optimally, screening should be performed by a speech and language therapist or other specially trained professional
- a standardized protocol is recommended
- implementation of oral hygiene protocols
- early nasogastric (NG) tube placement if dysphagia is evident, followed by swallowing training
- dietary modifications for milder cases
- if dysphagia persists beyond 2-3 weeks, percutaneous endoscopic gastrostomy (PEG) should be indicated (AHA/ASA 2019 IIa/C-EO)
- screening for dysphagia before initiating per os intake (including medications) – effective in identifying patients at increased risk of aspiration (AHA/ASA 2019 I/C-LD)
- screening tests
- GUSS test
- water swallow test (original and modified)
- the most common cause of GI bleeding is either a preexisting lesion or a newly developed “stress ulcer”
- disruption of the integrity of the upper GI mucosa due to extreme physiological stress, typically in critically ill patients
- often develops within a few hours after the initial insult
- can result in bleeding or perforation ⇒ ↑ mortality and intensive care stay
- incidence approx. 3% when on prophylactic medication
- risk factors for GI bleeding
- coagulopathies, including iatrogenic
- history of GI bleeding/peptic ulcer
- mechanical ventilation > 48h
- traumatic brain/spinal cord injury
- sepsis
- corticosteroids use
- renal and hepatic impairment
- malignancy
- severe stroke
- initiate enteral nutrition as soon as possible!
- prophylaxis should be administered only to patients at increased risk and discontinued in a timely manner (due to the increased risk of nosocomial pneumonia, Clostridium difficile infection, drug interactions, or hepatotoxicity); routine use of PPIs does not reduce mortality
- proton pump inhibitors (PPIs)
- PPIs are more expensive and significantly more effective than H2-blockers [Buendgens, 2016]
- use H2 blockers if PPIs are contraindicated
- 40 mg once daily, or 20 mg twice daily PO
-
- 1g PO or via nasogastric tube every 6-8 hours
- used in peptic ulcer prevention and treatment or to reduce hyperphosphatemia
- caused by a clonic contraction of the diaphragm with simultaneous closure of the glottis
- short-term hiccups are mostly benign and can be attributed to factors such as:
- distention of the esophagus and stomach, intake of carbonated fluids, irritation of the digestive tract with spices
- emotions, excitement
- sudden change in temperature: drinks (hot/cold), shower, air, etc.
- more serious underlying causes:
- pulmonary and mediastinal diseases (pneumonia, lung tumors, mediastinitis, and mediastinal tumors)
- abdominal cavity diseases (direct irritation of the diaphragm – ileus, peritonitis, stomach and liver tumors and metastases, liver abscess, pancreatitis, and pancreatic tumors, etc.)
- heart diseases (pericarditis, myocardial infarction)
- esophageal diseases (oesophageal obstruction by solid food or tumor, or esophagitis)
- metabolic causes (uremia, diabetes decompensation), acid-base disorders, mineral imbalances (hyponatremia)
- central causes (direct or indirect brainstem lesions) – tumors, stroke, trauma
- alcohol and drugs (dexamethasone, methyldopa, sulfonamides, antiseizure medications)
- severe forms of hiccups are frequently resistant to symptomatic treatment
- treat potential causes
- pharmacotherapy (see table) – combination therapy may be effective (e.g., omeprazole + baclofen + gabapentin)
- psychotherapy (cognitive-behavioral therapy or other psychological interventions may be beneficial for stress-induced hiccups)
- acupuncture (may provide symptomatic relief, particularly for hiccups resistant to pharmacotherapy)
| BACLOFEN (has a peripheral and central effect) |
|
| Anticonvulsive drugs |
|
| gabapentin (NEURONTIN) |
|
| valproate (ORFIRIL, DEPAKINE) |
|
| Neuroleptics (central effect) | |
| HALOPERIDOL |
|
| chlorpromazine (PLEGOMAZIN) |
|
| Prokinetic drugs |
|
| metoclopramide |
PO 10 mg every 6-8 hours (max 40 mg/d) |
| PPI (use if GER is suspected) | |
| omeprazole pantoprazole |
PO 20-40 mg once daily |
Urogenital complications
- urinary incontinence
- urinary tract infections (up to 15%)
- may lead to worsening of the clinical condition
- prevention:
- avoid unnecessary catheterization and regularly review the necessity of an indwelling catheter and remove it as soon as clinically feasible
- employ sterile technique during catheter insertion and maintenance
- use antibiotic-coated or silver-alloy catheters to reduce the risk of bacterial colonization
- perform routine urine tests to detect early signs of infection
- ensure adequate fluid intake to promote urinary flow and dilute bacteria
- encourage early mobilization
Deep vein thrombosis/pulmonary embolism
Other complications
- Poststroke Depression (PSD) is quite common (in up to 33% of patients)
- depressed patients are less compliant with physical therapy and medical treatment and have worse outcome compared to non-depressed patients
- early drug treatment (tricyclics, SSRIs, SNRIs) is important
- anxiety
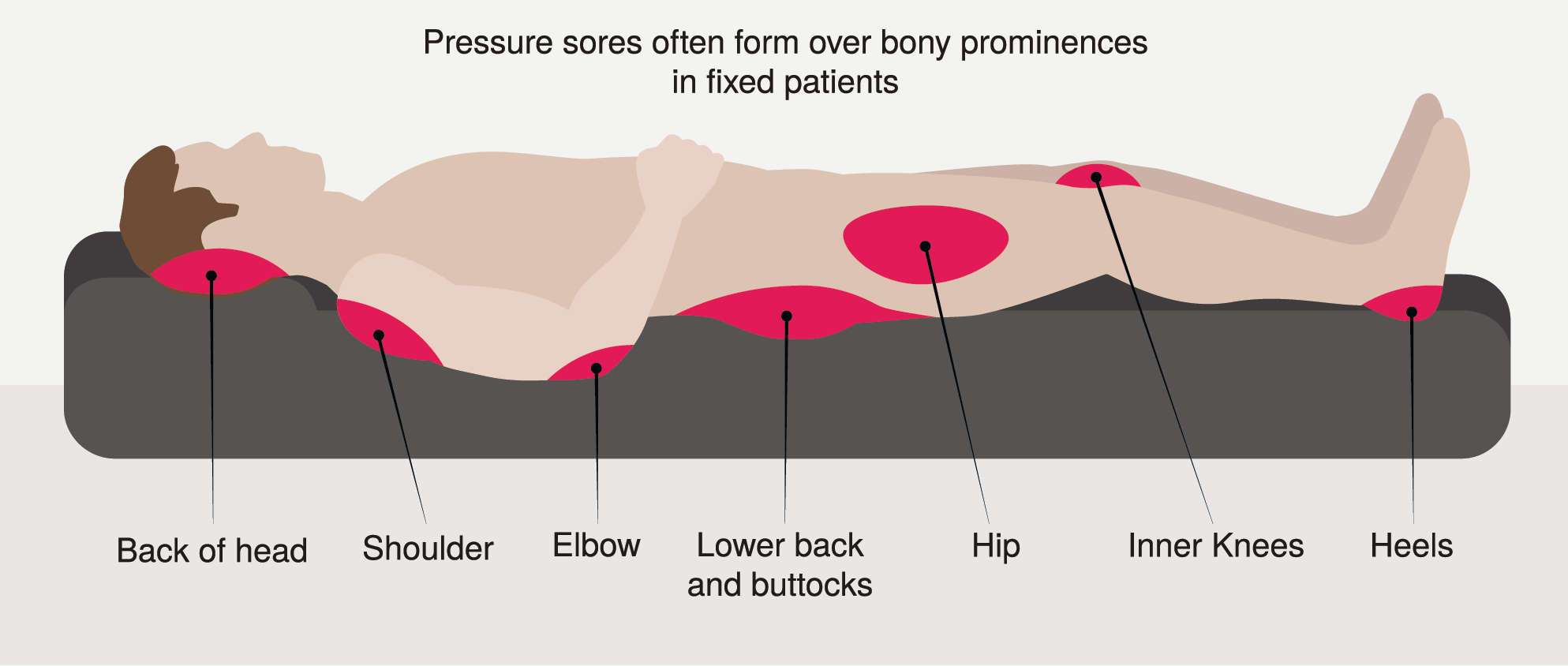
- pressure sores usually occur on the back of the head, shoulders, elbows, sacrum and buttocks, hips, and heels
- prevention of pressure sores:
- rule out fracture or luxation (often caused by a fall due to sudden onset of paresis)