SUBARACHNOID HEMORRHAGE
Superficial siderosis
David Goldemund M.D.
Updated on 11/03/2024, published on 06/04/2021
- cerebral superficial siderosis (SS) is a rare condition characterized by the deposition of hemosiderin (hemoglobin breakdown product) in the subarachnoid space (SA) on the surface of the brain and spinal cord, regardless of etiology
- its exact prevalence is not well-established due to its rarity and underdiagnosis
- occurs as a result of chronic or intermittent extravasation of blood into the SA space
- early diagnosis and treatment of dural defects are essential to reduce the risk of irreversible neurological injury
- on MRI, particularly on GRE/SWI sequences, it appears as a hypointense rim covering the surface of the brain and/or spinal cord
- the idiopathic progressive infratentorial form of SS (iSS) is a sporadic disease
 Classification
Classification
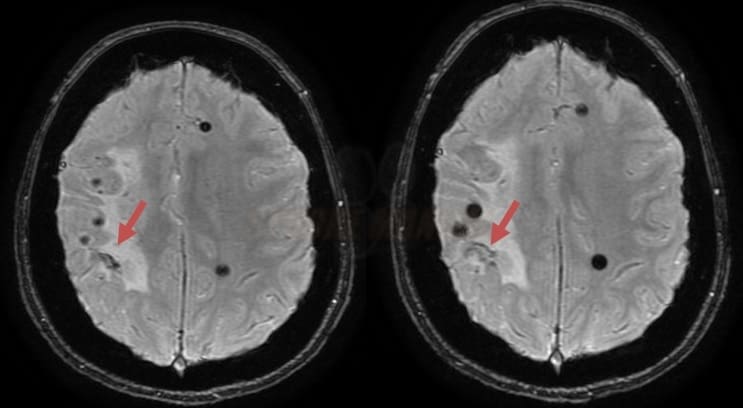
- cortical SS (cSS) → see also non-traumatic convexal SAH
- hemosiderin is localized in cortical sulci on convexities
- symptoms may include episodic and focal neurological symptoms related to the affected brain regions
- in older individuals, cSS is most commonly associated with cerebral amyloid angiopathy (CAA)
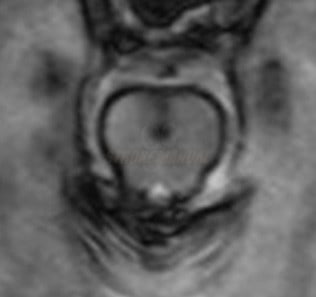
- infratentorial SS (iSS)
- involves the brainstem and cerebellum, with corresponding symptoms such as ataxia, myelopathy, and hearing impairment
- results from recurrent subarachnoid hemorrhage and deposition of hemosiderin in the subpial layers of posterior fossa structures
- forms:
- type 1 – idiopathic – patients with no apparent source or history of bleeding; most commonly attributed to spinal or cranial dural defects
- type 2 – secondary – patients with a history of prior intracranial hemorrhage, angioinvasive tumor, or vascular malformation
- type 1 – idiopathic – patients with no apparent source or history of bleeding; most commonly attributed to spinal or cranial dural defects
- involves the brainstem and cerebellum, with corresponding symptoms such as ataxia, myelopathy, and hearing impairment
 Pathophysiology
Pathophysiology
- cSS is most commonly attributed to focal convexal subarachnoid hemorrhage (SAH)
- iSS results from low-volume but prolonged or recurrent occult subarachnoid bleeding from altered intradural or epidural vessels
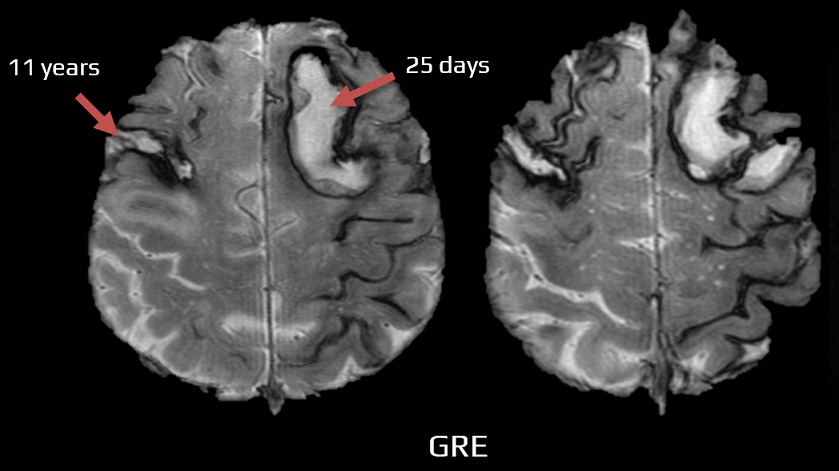
- a hemosiderin rim is often present at the edge of the hemorrhage or surgical resection; however, a single episode of bleeding rarely leads to significant symptoms – the condition requires repeated episodes of bleeding
- hemoglobin released from erythrocytes degrades into globin and neurotoxic heme in the cerebrospinal fluid (CSF). In response to the presence of heme, glial and microglial cells release heme oxygenase-1 and apoferritin. Subsequent heme catabolism yields free iron, whose surplus leads to neuronal injury
- trauma
- tumor – SS is caused by bleeding from a tumor or previous surgery
- vascular lesions
- cerebral and spinal AVMs
- spinal dural arteriovenous fistula (DAVF)
- aneurysm (including unruptured aneurysm)
- cavernous malformation [Hsu, 1999]
- cerebral venous thrombosis
- regrowing of fragile capillaries near the surgical field
- intracranial hemorrhage
- radiotherapy
- the most common cause of cSS in patients > 55 years of age is cerebral amyloid angiopathy (CAA)
- cSS with Transient Focal Neurological Episodes (TFNE) predicts an increased risk of ICH
- in younger patients, reversible cerebral vasoconstriction syndrome (RCVS) predominates
- rarely, cSS is caused by conditions whose acute manifestation may include convexal subarachnoid hemorrhage
- primary CNS angiitis (PACNS)
- bacterial endocarditis
- cerebral hyperperfusion syndrome
- PRES
- cerebral venous thrombosis
see chapter nontraumatic convexal SAH
- the source of bleeding often remains undetected (in up to 50% of cases)
- dural defects
- spinal and/or cranial
- sometimes associated with epidural fluid collection
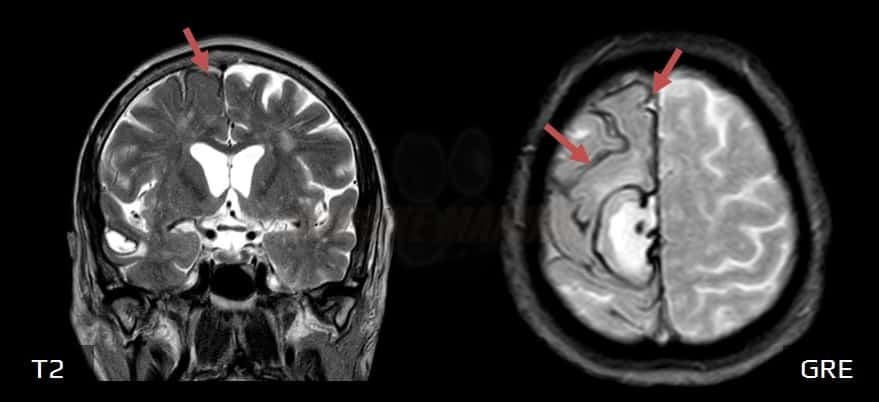
- postoperative pseudomeningocoele (an abnormal collection of CSF in the subcutaneous or extradural space following a dural breach during cranial/spinal surgery)
- dural ectasia (documented in conditions such as Marfan syndrome and neurofibromatosis type 1)
- craniospinal surgery
- the most common complication of posterior fossa surgery
- a dural tear, which may lead to the subsequent development of pseudomeningocele and SS
- development of SS has also been reported following ventriculoperitoneal shunt implantation
 Clinical presentation
Clinical presentation
- clinical presentation of SS varies depending on the localization and the extent of hemosiderin deposition
- may remain asymptomatic (incidental finding on MRI)
- the degree of clinical impairment may not correlate with the extent of imaging abnormalities
- distinguish the symptoms of the underlying disease that led to SS (e.g., dementia caused by CAA)
Infratentorial SS
- symptoms frequently appear several years after the onset of siderosis; symptoms and latency are probably related to the duration of bleeding and the amount of deposited hemosiderin
- gait ataxia
- myelopathy
- sensorineural hearing impairment (in up to 95% of cases)
- usually bilateral, asymmetric, and progressive
- may include both retrocochlear and cochlear components
- tinnitus
- impairment results from a long cisternal course of the nerve and the susceptibility of microglial cells to iron compounds [Swartz, 2008]
- dysphagia or cerebellar dysarthria may occur in advanced stages, potentially leading to misdiagnosis of degenerative cerebellar ataxia
Cortical SS
- epileptic seizures (< 5% of cases)
- cognitive deterioration in widespread cSS
- sensory deficits
- cranial neuropathies
- anosmia
 Diagnostic evaluation
Diagnostic evaluation
- brain CT + CT angiography
- excludes structural or vascular pathology
- identifies atrophy or posttraumatic changes in parenchyma or bones
- may show a hyperdense rim around the brainstem
- MRI of the brain and spinal cord
- the modality of choice to demonstrate hemosiderin deposition
- T1 – hypointense rim
- T2 – hypointense rim mainly on the surface of the cerebellum (vermis and folia), brainstem, and spinal cord, often also in the area of the Sylvian furrow or cerebral convexity (however, secondary gliosis can also cause increased signal)
- GRE/SWI – hypointense lesions with blooming artifacts in the same location
- often symmetric and confluent hypointensity following the contours of the brain and spinal cord
- iSS typically affects the cerebellum and brainstem, most commonly the superior vermis or vestibulocochlear nerve (CN VIII)
- T1 – hypointense rim
- in the classic form of iSS, cerebellar atrophy is present, mainly affecting the superior vermis
- exclude myxopapillary ependymoma among spinal cord lesions
- the modality of choice to demonstrate hemosiderin deposition
- CT or MR myelography
- epidural fluid collection
- dural defect
- CSF analysis
- potential evidence of ongoing bleeding (such as xanthochromia and elevated protein levels)
- CSF may normalize with a longer interval since the last attack
- cerebral or spinal DSA
- rarely identifies an apparent source of bleeding not seen on CTA/MRA
- rarely identifies an apparent source of bleeding not seen on CTA/MRA
- in up to 50% of cases, no source of bleeding can be identified (but a history of head trauma or surgery is often present)
 Management
Management
Causal therapy
- elimination of the source of bleeding, if identified
- dural defects can be sealed via sutures, patches, or fibrin glue
- even with successful surgical treatment, clinical progression may not be halted
- a secondary neurodegenerative process may be the reason for treatment failure
- pharmacotherapy is generally ineffective (including corticosteroids, selegiline, vitamins C and E)
- promising results have been published from a pilot study with (an iron chelator) in which 50% of patients showed MRI improvement and stabilization of neurological deficits
[Kesseler, 2017]
- may cause adverse effects such as agranulocytosis or hepatic dysfunction, requiring regular monitoring
Symptomatic therapy
- most symptoms of infratentorial SS, such as ataxia or myelopathy, cannot be effectively treated
- supportive therapy can help patients adapt and minimize further neurological disabilities ⇒ physical and occupational therapy
- consider a cochlear implant for sensorineural hearing loss