SUBARACHNOID HEMORRHAGE
Management of asymptomatic intracranial aneurysm
David Goldemund M.D.
Updated on 29/04/2024, published on 25/11/2022
- the prevalence of unruptured intracranial aneurysms (UIA) is ~ 0.5-5%
- modern and widely used imaging methods increase the rate of detection of incidental intracranial aneurysms
- multiple aneurysms are found in 20-30% of cases
- modern and widely used imaging methods increase the rate of detection of incidental intracranial aneurysms
- management of unruptured asymptomatic aneurysms is controversial
- the decision-making process involves weighing the individual risks of conservative management versus intervention, considering several prognostic factors
- distinguish:
- incidental aneurysm + personal history of SAH from another aneurysm – the highest risk of bleeding (up to 10-fold compared to true incidental aneurysm)
- incidental aneurysm + positive family history of SAH
- incidental aneurysm + negative family and personal history of SAH
 Risk factors
Risk factors
| Risk factors for aneurysm formation | |
| Congenital | Acquired |
| AD polycystic kidney disease Fibromuscular dysplasia (MD) hereditary hemorrhagic telangiectasia neurofibromatosis type 1 tuberous sclerosis coarctation of the aorta Ehler-Danlos type IV Pheochromocytoma Marfan syndrome α1-antitrypsin deficiency |
age > 50 years smoking alcohol abuse cocaine or amphetamine abuse atherosclerosis head trauma |
 Prognostic factors
Prognostic factors
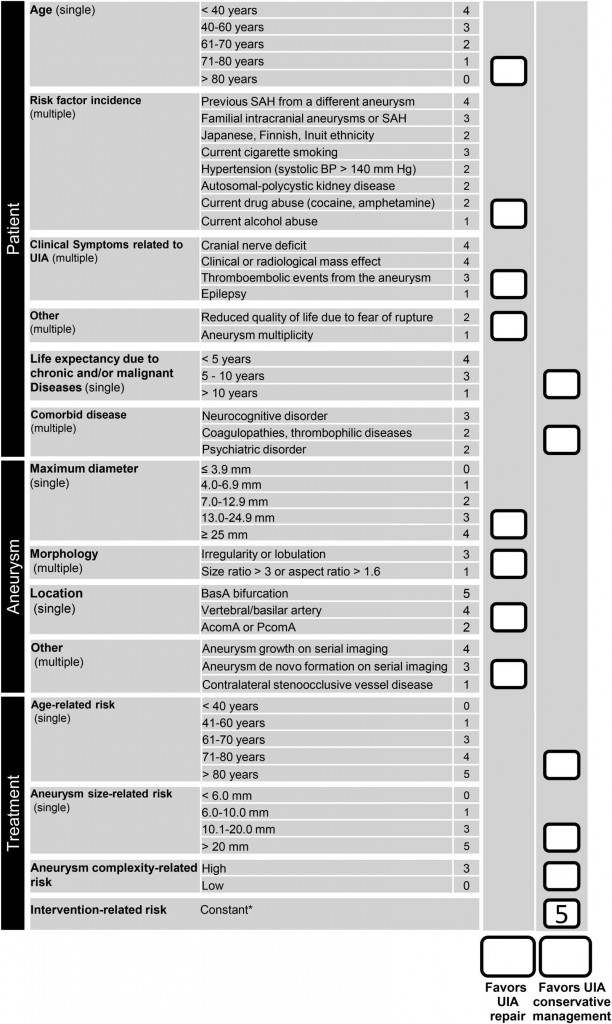
- several factors significantly influence surgical outcome, and these factors should be considered when evaluating treatment alternatives (AHA guidelines 2020)
- these factors can be grouped into patient characteristics (age, symptoms, and medical condition), aneurysm characteristics (size, location,
and morphology), and other factors (hospital and surgical team experience)
Aneurysm characteristics
Patient characteristics
- age, gender, and comorbidities (⇒ higher surgical risk)
- according to ISUIA, the mortality/morbidity rate was 6.5% for patients < 45 years, 14.4% for patients 45-65 years, and 32% for those over 65 years
- ↑ risk of rupture in decompensated hypertension
- ↑ risk for smokers (up to 4-fold in women) [Ogilvy, 2020]
- ↑ risk in women
- according to ISUIA, the mortality/morbidity rate was 6.5% for patients < 45 years, 14.4% for patients 45-65 years, and 32% for those over 65 years
- race
- history of SAH due to another aneurysm (~10-fold risk)
- family history of SAH (highest risk with ≥ 2 relatives are affected)
- the chance of having an aneurysm correlates with the number of affected first-degree relatives (higher risk for siblings rather than parents or children)
- aneurysms in familial SAH cases are more frequently large and multiple compared to sporadic cases
- patients with familial SAH tend to be younger than those with sporadic cases
Experience of a surgeon/interventional radiologist
- combined morbidity/mortality
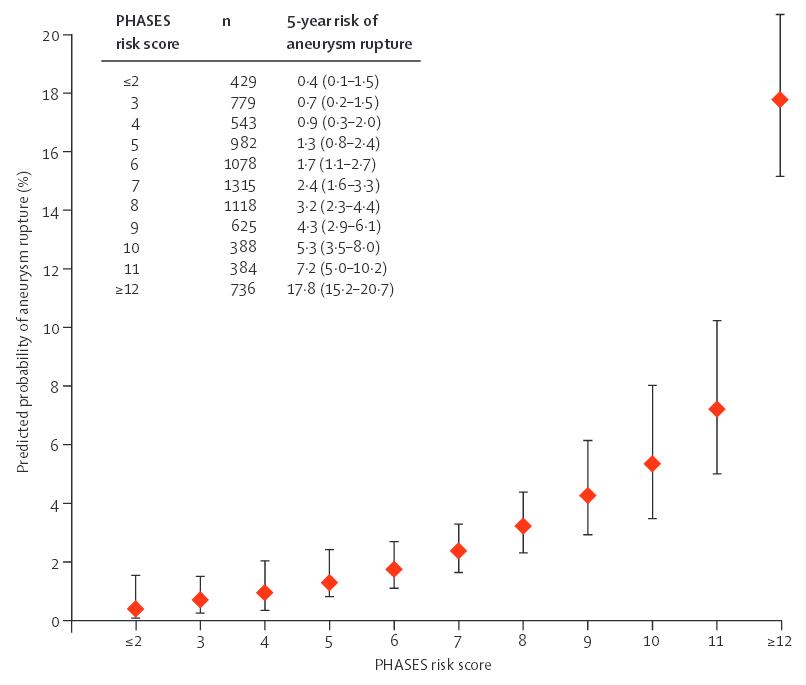
Prognostic scores
 Management
Management
- patients should be informed about the risks and benefits of conservative treatment, clipping, and coiling (AHA/ASA 2009 IIa/B)
- procedure risks:
- post-craniotomy epilepsy
- periprocedural stroke
- aneurysm rupture
- wigh the risk of rupture per year against the patient’s life expectancy
- a randomized trial comparing surgical and conservative management of unruptured aneurysms is not available and cannot be expected in the neart future
- there is a paradox between the fact that, according to ISUIA, the risk of bleeding is low for small aneurysms (<7 mm ⇒ 0.7% per year) and the observation that most bleeding aneurysms in routine practice are in the 7-10 mm range
- it is assumed that some aneurysms undergo a period of higher risk of rupture after their formation, followed by stabilization and transition to a period of low risk – some of these aneurysms are likely to bleed shortly after their formation
- therefore, it makes sense to treat only incidental aneurysms that grow or change their shape
- long-term stable aneurysms have a low risk of bleeding
ISUIA (International Study of Unruptured Intracranial Aneurysms) study is the largest and most widely discussed trial
- n= 4060, three groups: no surgery x clipping x coiling
- the larger the aneurysm, the greater the risk of rupture
- higher risk for aneurysms in PComA, PCA, and basilar artery
- higher risk in patients with previous SAH from another source (approx. 10-fold)
- the prospective arm (1991-1998) had an overall higher incidence of bleeding than the retrospective arm (1970-1991) – 0.8% vs. 0.3%
- according to the prospective data, the risks of clipping and coiling are comparable, but coiling achieved complete obliteration in only 51% of cases
- patients with aneurysms in the anterior circulation (MCA, AComA) had better outcomes when they underwent clipping rather than coiling
- the differences in the results between the retrospective and prospective arms make the generalization of the results problematic
- some authors believe that the ISUIA study underestimates the risk of bleeding, as most ruptured aneurysms fall within the 7-10 mm diameter range
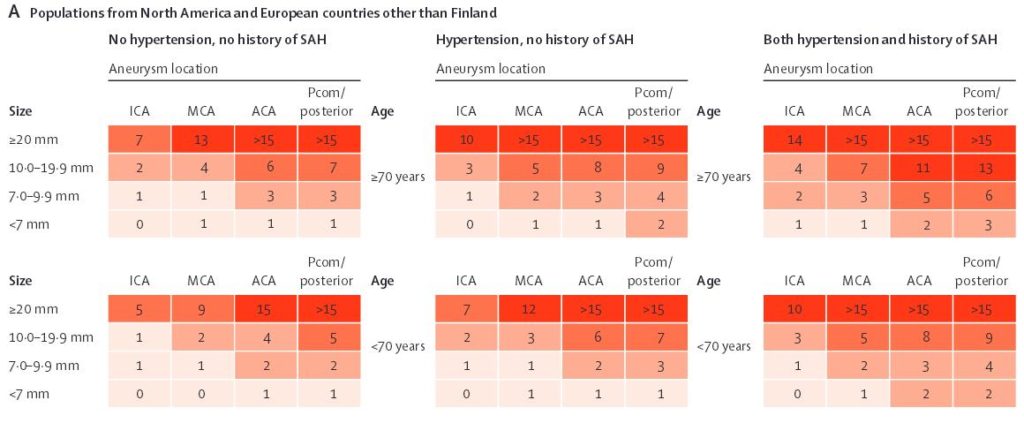
| The 5-year cumulative risk of rupture | ||
| anterior circulation |
posterior circulation (incl. PCoA) | |
| < 7 mm | 0% | 2.5% |
| 7-12 mm | 2.6% | 14.5% |
| 13-24 mm | 14.5% | 18.4% |
| ≥ 25 | 40% | 50% |
A rather conservative approach
- unruptured, asymptomatic aneurysms < 7mm (especially in the anterior circulation) with no history of SAH
- low PHASES score
- UIA score favoring conservative management
- older age and severe comorbidities
- for large asymptomatic intra-cavernous ICA aneurysms, management is individualized
- monitor patients with 3D TOF MRA → procedure is recommended if aneurysm growth or daughter aneurysm formation is detected
Rather surgical/interventional procedure
| Content available only for logged-in subscribers (registration will be available soon) |
 Follow-up
Follow-up
- the first follow-up (MRA/CTA) should be done in 6-12 months after the procedure, then every 1 or 2 years
 Screening for asymptomatic aneurysms
Screening for asymptomatic aneurysms
- generally not recommended (only grade C recommendations; cost-effectiveness is unknown)
- screening is not recommended in the general population, including smokers and alcoholics (AHA guidelines 2020)
- may be considered in female smokers aged 30-60 years [Ogilvy, 2020]
- no indication for patients with Ehlers-Danlos syndrome type IV (high risk of treatment), neurofibromatosis, or Marfan syndrome
- screening with noninvasive methods is recommended for:
- patients with a history of aneurysmal SAH (↑ risk of new aneurysm formation)
- first-degree relatives of patients with aneurysmal SAH (especially siblings)
- individuals with autosomal dominant polycystic kidney disease (ADPKD)
- choice of screening method
- MRA is preferred (no contrast injection and radiation exposure)
- both CTA and MRA have a risk of missing very small aneurysms (which are typically not treated)
- in patients with previously clipped aneurysms, CTA is preferred (extensive artifacts caused by clips on MRA)
- MRA is preferred in patients who have undergone coiling (as coils produce minimal artifacts on MRA)
- more data are needed to help identify patients who would benefit from screening
Development of new aneurysms
- intracranial aneurysms are not congenital lesions but develop (grow) during life
- patients who have survived an episode of SAH are at increased risk of having a new bleeding episode from:
- treated aneurysm
- already existing, known aneurysm
- new aneurysm
- the annual rate of new aneurysm formation in patients treated for aneurysmal SAH is reported 1-2%
- risk factors include multiple aneurysms at the time of SAH, smoking, and hypertension
- recurrent or de novo aneurysms are found in up to 30% of patients with SAH ⇒ screening should be repeated in a high-risk population with negative baseline imaging